Benzene
Metabolism of benzene and its relevance to benzene carcinogenicity
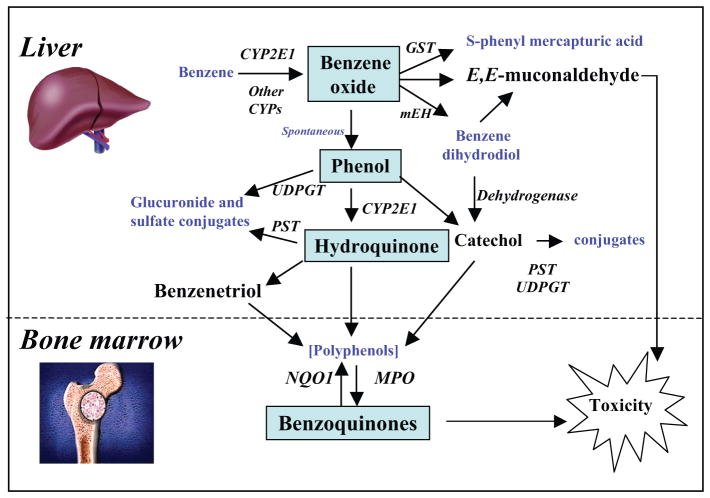
Benzene must be metabolized to become carcinogenic. Its metabolism is summarized in figure. The initial step involves cytochrome p450 (cyp)-dependent oxidation of benzene to benzene oxide, which exists in equilibrium with its tautomer oxepin. Most benzene oxide spontaneously rearranges to phenol (ph), which is either excreted or further metabolized to hq and 1,4-bq. The remaining benzene oxide is either hydrolyzed to produce catechol (ca) and 1,2-bq or reacts with glutathione to produce s-phenylmercapturic acid (spma). Metabolism of oxepin is thought to open the aromatic ring, yielding the reactive muconaldehydes and e,e-muconic acid (ma). Human exposures to benzene at air concentrations between 0.1 and 10 ppm result in urinary metabolite profiles with 70%–85% ph, 5%–10% each of hq, ma, and ca, and less than 1% of spma. Benzene oxide, the bqs, muconaldehydes, and benzene diol epoxides (formed from cyp oxidation of benzene dihydrodiol) are electrophiles that readily react with peptides and proteins and can thereby interfere with cellular function. It remains unclear what role these different metabolites play in benzene carcinogenicity, but bq formation from hq via myeloperoxidase in the bone marrow may be key. Considerable evidence indicates that this pathway plays an important role in bq formation because the bq-detoxifying enzyme nqo1 protects mice against benzene-induced myelodysplasia and protects humans against benzene hematotoxicity. However, this protection does not rule out adverse effects from other metabolites.
Figure 2
Simplified metabolic scheme for benzene showing major pathways and metabolizing enzymes leading to toxicity. CYP2E1, cytochrome P450 2E1; GST, glutathione-S-transferase; NQO1, NAD(P)H:quinone oxidoreductase 1; MPO, myeloperoxidase; UDPGT, uridine diphosphate glucuronyl transferase; PST, phenol sulfotransferase; mEH, microsomal epoxide hydrolase.